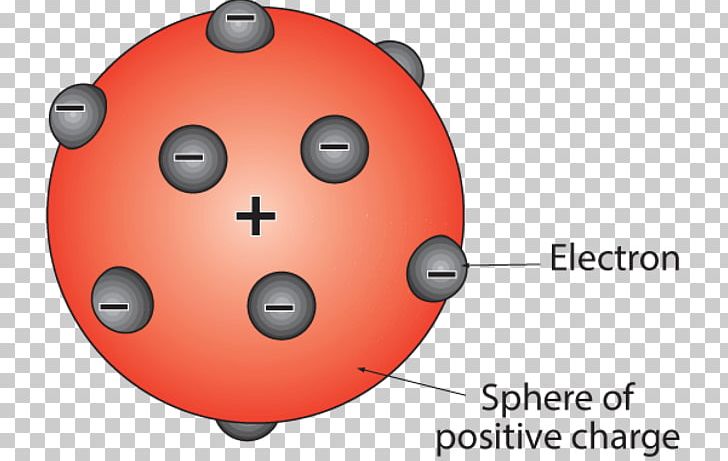 
The electron does not have a definite location but instead exists as a probability cloud.\): Bohr models of different groups/families of elements from the periodic table. The concept that matter is composed of discrete particles is an ancient idea, but gained scientific credence in the 18th. JJ Thomson Proposed that an atom consists. The electrons were assumed to be positioned in revolving circles around the atom in this model to be having a 'cloud' of positive charge. plum pudding model positive charge spread throughout the atom (1) accept. 
Atomic theory is the scientific theory that matter is composed of particles called atoms. Thomson, who invented the electron in the year 1897, suggested the atom's plum pudding model in 1904 which was for including the electron in the atomic model. The diagram is labelled 'Plum Pudding Atomic Model. 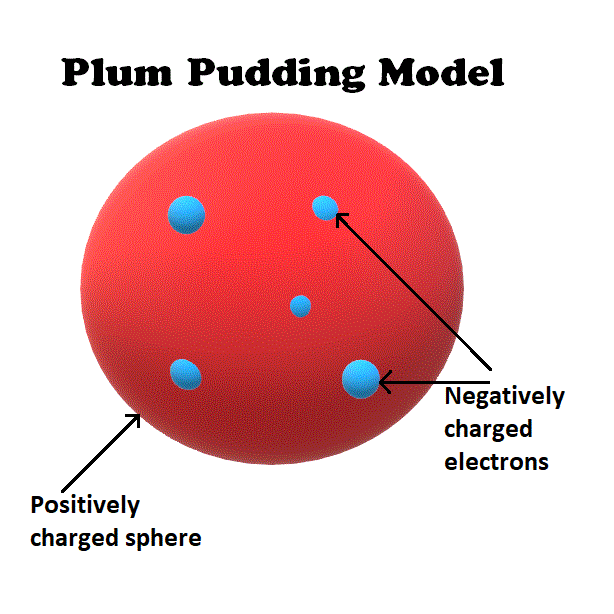
The golden brown area is labelled 'Cake', and the dark ovals are labelled 'Raisins.' On top is a sprig of holly with two green leaves and three red berries. Another limitation is that the Thomson model does not account for the electron’s quantum nature. This discovery was an atomic model in which the interior of the atom was uniformly positively charged with negatively charged electrons spread throughout it. The current theoretical model of the atom involves a dense nucleus surrounded by a probabilistic 'cloud' of electrons. On the left, the photograph is labelled 'Plum pudding.' It shows a golden brown, dome-shaped cake with dark brown, oval shapes throughout. Thomson believed that the nucleus was composed of positive particles, but it is now known that the nucleus is composed of protons and neutrons. Proposed in 1904, it suggested that atoms were composed of negatively charged electrons embedded in a pudding of positive charge, much like plums in a pudding. Since the overall atom is neutral, the plums (electrons) would be negatively charged and the pudding (protons) would be positively charged. In this atomic model, electrons were viewed as being small particles embedded in the atom, much like a plum in a bowl of pudding. One is that it does not account for the structure of the nucleus. This video will teach you about the plum pudding model. His two students, Hans Geiger and Ernest Marsden, directed a beam of alpha particles close alpha particle Subatomic. The Thomson model has a number of limitations. The name stuck (like a plum pudding does sometimes) and it became the accepted model around the start of the 20th century. In 1905, Ernest Rutherford did an experiment to test the plum pudding model. Thomson believed that the positive charge of the nucleus was due to the presence of positive particles, which he called corpuscles. In this model, the atom is composed of a positively charged, central nucleus surrounded by a negatively charged, electron cloud. Thomson’s model of the atom is also called the plum pudding model because of its resemblance to a plum pudding. The plum pudding model The Geiger-Marsden experiment (1909 - 1911) Hans Geiger and Ernest Marsden tested the plum pudding model. Limitations of JJ Thomson Model of Atom (Plum Pudding Model) The plum pudding model was eventually replaced by the Rutherford model, which was developed in 1911 by Ernest Rutherford. Thomson did not know what held the electrons in their orbits, but he guessed that it was some sort of force field. Thomson’s model consisted of a positively charged sphere in the center of the atom, with the electrons orbiting around it like planets orbiting around the sun.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
